CTD
Course Objectives
1- Explain the history and need for CTD as an international regulatory working standard.
2- Clarify the granularity of CTD format dossiers according to ICH guidance.
3- Illustrate the scientific data needed to complete each part & create a strong CTD file submission.
4- To train the participants to plan and prepare a successful CTD submission.
Course Outline
► Overview and history of CTD and e-Submissions.
CTD advantages against different types of submissions.
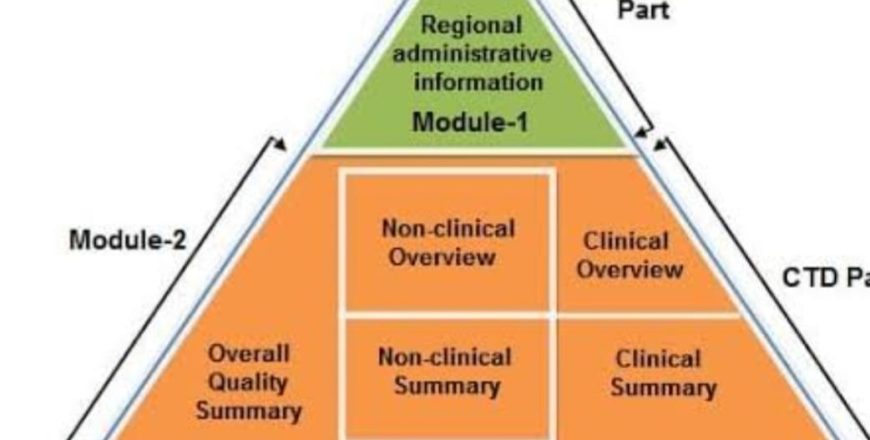
► CTD format outline (modules 1 – 5)
CTD submission workflow inside and outside the corporate.
CTD Module (1 – 5) in detail.
►Hard copy specifications and management.
CTD Example for registration in EDA. (Module 3)
1
Introduction
2
Lec1
3
Lec2
4
Lec3
5
Lec4
